1. In the shadows: alkali

Like an under-performing sibling, alkali is mentioned way less than acid (view graph above). But it is no less interesting. In this set of notes, we will explore its properties and reactions with ammonium salt and acid. We will also consider a probable reason why the term alkali is used way less: it is subsumed under a bigger group of chemicals called bases.
2. In water: alkali forms hydroxide ions
| Common Alkalis | Chemical Formulae |
|---|---|
| sodium hydroxide | NaOH |
| potassium hydroxide | KOH |
| calcium hydroxide | Ca(OH)2 |
| ammonia | NH3 |
Alkalis come in all forms. They have very different chemical compositions revealed by their chemical formulae. They have different bonding: some like sodium hydroxide are ionic compounds while ammonia is a simple molecule. However, they are united and defined by what they do in water.
Alkalis are compounds that dissolve in water to form hydroxide ions, OH–.

The hydroxide ions produced in water explain why dissolved alkalis:
- Have a pH greater than 7, hence turning Universal Indicator blue or violet
- Turn moist red litmus paper blue
- Conduct electricity as aqueous hydroxide ions are free-moving
3. One way or another: forming hydroxide ions by dissociation or reaction
Dissociation of Soluble Metal Hydroxides
NaOH(s) ⟶ Na+(aq) + OH–(aq)
Soluble metal hydroxides produce hydroxide ions in water by the simple process of dissociation. Take sodium hydroxide as an example. In the solid state, the sodium cations and hydroxide anions are held together by ionic bonds. However, when a solid pellet of sodium hydroxide dissolves in water, the bonds are overcome to free up the hydroxide ions. We call this process dissociation.
Reaction of Ammonia with Water
Ammonia is a tricky alkali. When you inspect its molecular formula of NH3, you do not see any remnant of hydroxide. However, there is more than meets the eye.
NH3(g) + H2O(l) ⇄ NH4+(aq) + OH–(aq)
Ammonia does not just dissolve in water. It also reacts with water, stripping water of a hydrogen to produce hydroxide ions alongside ammonium ions, NH4+. This reaction is also called ionisation, as simple molecules of ammonia and water react to form ions.
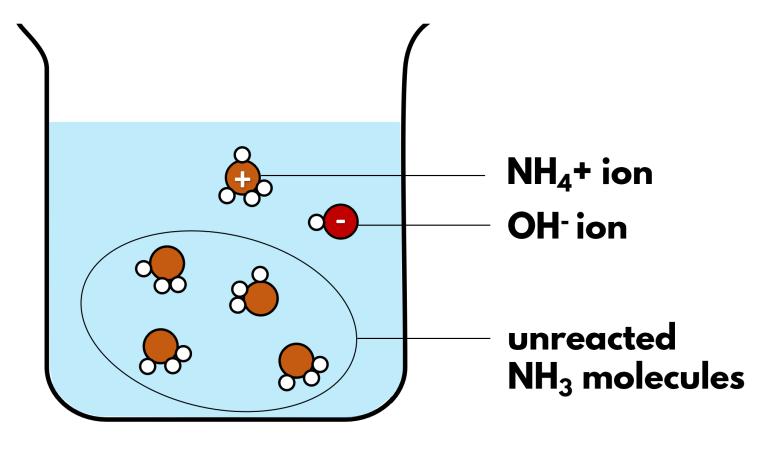
The reaction between ammonia and water is partial, as indicated by the double arrow in the ionic equation. In other words, if you look into a solution of aqueous ammonia, only a small fraction of the ammonia molecules have ionised to form ammonium ions and hydroxide ions. Most ammonia remains as simple molecules in the solution. Therefore, we say that ammonia is a weak alkali that undergoes partial ionisation.
4. Alkali reacts with ammonium salt to release ammonia gas
Dissolved ammonia, NH3 takes a hydrogen ion from water to form ammonium ion, NH4+. The same ammonium ion is found in ammonium salts like ammonium chloride, ammonium nitrate, and ammonium sulfate.
NaOH(aq) + NH4Cl(aq) ⟶ NaCl(aq) + H2O(l) + NH3(g)
The hydroxide ions of an alkali can react with the ammonium ions of these ammonium salts. During the reaction, hydroxide ions will snatch a hydrogen back from ammonium ions, reforming ammonia and water. For example, the alkali sodium hydroxide reacts with ammonium chloride to form sodium chloride, water, and ammonia gas.
Alkali reacts with ammonium salt to form another salt, water, and ammonia gas.
5. Alkali reacts with acid to form salt and water

We can see the hydroxide ions of alkali as modified water with a missing hydrogen. They can reform water when they combine with the hydrogen ions of an acid. Acid-alkali neutralisation gives a salt as a side product, which is formed by the cation of the alkali and the anion of the acid.
NaOH(aq) + HCl(aq) ⟶ NaCl(aq) + H2O(l)
For example, sodium hydroxide undergoes neutralisation with hydrochloric acid to form sodium chloride and water. The salt, sodium chloride, got its sodium cation from the alkali, and the chloride anion from the acid.
The Special Case of Ammonia
Ionisation: NH3(aq) + H2O(l) ⇄ NH4+(aq) + OH–(aq)
Neutralisation: NH4+(aq) + OH–(aq) + HCl(aq) ⟶ NH4Cl(aq) + H2O(l)
Overall equation: NH3(aq) + HCl(aq) ⟶ NH4Cl(aq)
Interestingly, when aqueous ammonia reacts with an acid, only an ammonium salt is formed. For example, aqueous ammonia reacts with hydrochloric acid to form ammonium chloride. You don’t see water in the equation, because the water produced in neutralisation is “cancelled out” by the water that has been used up during ionisation.
Alkali undergoes neutralisation with acid to form salt and water.
6. Get salty with alkali and acidic oxides
| Acidic Oxide | Reaction with Alkalis |
|---|---|
| carbon dioxide | CO2(g) + Ca(OH)2(aq) ⟶ CaCO3(s) + H2O(l) |
| sulfur dioxide | SO2(g) + 2NaOH(aq) ⟶ Na2SO3(aq) + H2O(l) |
| silicon dioxide | SiO2(s) + 2NaOH(aq) ⟶ Na2SiO3(aq) + H2O(l) |
Most non-metal oxides — with the exception of water, carbon monoxide, and nitrogen monoxide — are acidic oxides. Alkalis react with acidic oxides to form salt and water.
In fact, this reaction happens when we bubble carbon dioxide into limewater! Limewater is an alkaline solution of calcium hydroxide, Ca(OH)2. It reacts with carbon dioxide to form calcium carbonate and water. As calcium carbonate is an insoluble salt, it crashes out as a white precipitate.
7. But non-alkalis can also undergo neutralisation
Cu(OH)2(s) + H2SO4(aq) ⟶ CuSO4(aq) + 2H2O(l)
CuO(s) + H2SO4(aq) ⟶ 2CuSO4(aq) + H2O(l)
Neutralisation is not exclusive to alkalis. Insoluble metal hydroxides and metal oxides can also undergo neutralisation with acid to form salt and water. For example, insoluble copper(II) hydroxide reacts with sulfuric acid to form copper(II) sulfate salt and water. Insoluble copper(II) oxide can also react with sulfuric acid to give the same products.
8. A big family of compounds that neutralise acids
| Types of Bases | Examples |
|---|---|
| alkalis | sodium hydroxide, NaOH potassium hydroxide, KOH ammonia, NH3 |
| insoluble hydroxides* | aluminium hydroxide, Al(OH)3 lead(II) hydroxide, Pb(OH)2 iron(II) hydroxide, Fe(OH)2 copper(II) hydroxide, Cu(OH)2 |
| metal oxides | calcium oxide, CaO iron(II) oxide, FeO zinc oxide, ZnO |
Since there are compounds other than alkalis that react with acids, a new category of compounds is birthed: bases. It is a big reunion of alkalis, insoluble hydroxides and insoluble metal oxides. Their defining trait is the ability to neutralise acids to produce salt and water only.
This definition excludes carbonates and ammonium salts. While acid-carbonate and alkali-ammonium reactions do form salt and water, carbon dioxide and ammonia gas are also given off as by-products.
A base is a compound that undergoes neutralisation with acid to form salt and water only.
As base is an all-encompassing category, the term base is more commonly used. This may explain why the term alkali is so infrequent relative to the term acid.
9. The curious but useful case of calcium hydroxide
Calcium hydroxide treads the boundary of alkali. It is sparingly soluble. In other words, only small fraction of calcium hydroxide can partially dissolve in water to form a mildly alkaline solution. When the concentration increases beyond the maximum, any additional calcium hydroxide cannot dissolve and remains as a solid.
This gentle nature of calcium hydroxide makes it perfect to nurture plants. To treat acidity in soil, farmers and gardeners add calcium hydroxide solution, which is also called slaked lime. This is essential for plant survival, as very low pH would reduce the availability of mineral salts. It is also argued to be important for taste. Wine producers grow grapes in specific pH to enhance their flavours.
10. Some metal oxides react with both alkalis and acids
| Basic Oxides | Amphoteric Oxides |
|---|---|
| sodium oxide, Na2O potassium oxide, K2O magnesium oxide, MgO calcium oxide, CaO iron(II) oxide, FeO copper(II) oxide, CuO |
aluminium oxide, Al2O3 zinc oxide, ZnO lead(II) oxide, PbO |
While all metal oxides react with bases, there are three odd balls that also react with acids: zinc oxide, lead(II) oxide, and aluminium oxide. They are called amphoteric oxides. While amphoteric looks cheem, it comes from the Greek word ampho that means both.
Those that react with bases only are the basic oxides.


